SARS-CoV-2 SEROLOGICAL TESTS
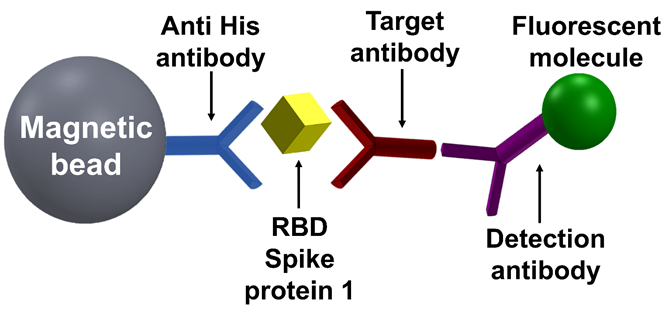 The gold standard for the diagnosis of coronavirus disease 2019 (COVID-19) uses the reverse-transcription quantitative polymerase chain reaction (RT-qPCR) to directly detect the virus’ ribonucleic acid (RNA) in nasopharyngeal swab samples. While RT-qPCR is accurate, its diagnostic efficacy is limited to a few weeks after the infection. Serological assays to detect antiviral antibodies against SARS-CoV-2, namely immunoglobulin M (IgM), immunoglobulin G (IgG), and immunoglobulin A (IgA), are not limited to the first weeks of the disease and they indicate previous or recent SARS-CoV-2 infection, irrespective of whether the individual had a severe, mild, or even asymptomatic infection. IgA and IgM antibodies typically rise several days after SARS-CoV-2 infection, remain high for ~7–14 days, and indicate an acute viral infection. IgG antibodies are usually detected ~14 days after infection, remain detectable for several months, and indicate past viral infection. The study presents a rapid, highly sensitive, and specific anti-SARS-CoV-2 IgG serological assay based on the MMB technology. Compared with the gold standard ELISA test, the MMB-based assay demonstrates an ~3.8–6-fold better limit of detection. In clinical tests, the MMB-based assay showed similar sensitivity (93% vs. 92%) and specificity (98% vs. 99%) as the ELISA test but managed to correctly detect 14 positive samples (56%) in a group of 25 RT-qPCR SARS-CoV-2-positive samples taken from a large cohort study and had been falsely identified as negatives using the ELISA test. In addition, it detected an increase in IgG antibody concentrations in vaccinated individuals much earlier with a much faster turnaround time (45 vs. 245 minutes).
The gold standard for the diagnosis of coronavirus disease 2019 (COVID-19) uses the reverse-transcription quantitative polymerase chain reaction (RT-qPCR) to directly detect the virus’ ribonucleic acid (RNA) in nasopharyngeal swab samples. While RT-qPCR is accurate, its diagnostic efficacy is limited to a few weeks after the infection. Serological assays to detect antiviral antibodies against SARS-CoV-2, namely immunoglobulin M (IgM), immunoglobulin G (IgG), and immunoglobulin A (IgA), are not limited to the first weeks of the disease and they indicate previous or recent SARS-CoV-2 infection, irrespective of whether the individual had a severe, mild, or even asymptomatic infection. IgA and IgM antibodies typically rise several days after SARS-CoV-2 infection, remain high for ~7–14 days, and indicate an acute viral infection. IgG antibodies are usually detected ~14 days after infection, remain detectable for several months, and indicate past viral infection. The study presents a rapid, highly sensitive, and specific anti-SARS-CoV-2 IgG serological assay based on the MMB technology. Compared with the gold standard ELISA test, the MMB-based assay demonstrates an ~3.8–6-fold better limit of detection. In clinical tests, the MMB-based assay showed similar sensitivity (93% vs. 92%) and specificity (98% vs. 99%) as the ELISA test but managed to correctly detect 14 positive samples (56%) in a group of 25 RT-qPCR SARS-CoV-2-positive samples taken from a large cohort study and had been falsely identified as negatives using the ELISA test. In addition, it detected an increase in IgG antibody concentrations in vaccinated individuals much earlier with a much faster turnaround time (45 vs. 245 minutes).
Our paper on SARS-CoV-2 Serological tests was published at "Biosensors". The paper, entitled: "Highly Sensitive and Specific SARS-CoV-2 Serological Assay Using a Magnetic Modulation Biosensing System", can be found in the Publications section, and at DOI:10.3390/bios12010007.
